Dew point vs condensation temperature
As natural gas infrastructure continues to expand and as more and more entities become involved with the extraction, processing and delivery of natural gas, quality control becomes increasingly critical. Historically, natural gas tended to be supplied from a single source, so once the specific characteristics of that gas were known, relatively few continuous quality control measurements were deemed necessary. But now, as the interconnectivity of the world’s natural gas infrastructure is advanced, and more gas is delivered as LNG via ship, the gas mixture in the pipeline can change significantly from one day to the next. It is largely for this reason that continuous automated quality control measurements have become the norm. But what exactly do we mean by quality control when it comes to natural gas?
Quality control technology
One aspect of quality control is to measure various aspects of the gas. Natural gas quality is measured in terms of calorific value and Wobbe index, moisture content and hydrocarbon condensation temperature (HCT), as well as several other parameters. Part of the challenge to good quality control is the fact that natural gas is different things at different points in the delivery chain and, in all cases, it is a complex gaseous media comprising multiple components. Some aspects of the gas quality are inherent in each of these components, so modern quality control practices require appropriate monitoring – often if only to ensure that a given component is present in no more than trace amounts.
A number of technologies have been developed to accurately evaluate different aspects and components of the gas. Of these, the two measurements that are often the most essential are the water dew point (WDP) and the HCT. Perhaps because the HCT has traditionally been referred to as the hydrocarbon ‘dew point’, there is often confusion regarding the differences and similarities of these measurements. In some cases, apparently because natural gas is mostly hydrocarbons, the misconception has arisen that the ‘hydrocarbon dew point’ is just the name for the WDP in a hydrocarbon gas; this is most definitely not so.
Water dew point
To better understand the difference between the WDP and the HCT, let us examine each measurement in turn, beginning with the WDP.
One of the first quality-related measurements involved determining the water content of natural gas. The presence of excess moisture in the gas leads to a number of adverse situations. Not only does moisture reduce the actual energy in the gas for a given volume, it can also lead to the formation of clathrate hydrates (also known as gas clathrates, gas hydrates, clathrates, etc.). These are ‘slushy’ ice-like solids formed of water and methane. These hydrates can create blockages that restrict or completely clog pipelines.
In addition to clathrates, excess water can combine with CO2 to form the aggressive compound carbonic acid, which can cause corrosive damage to pipes and equipment. Water can likewise interact with excess hydrogen sulfide (H2S) to form an acidic compound. And of course, water can cause ordinary rust. So, for all these reasons and more, steps are taken to remove water from the gas at an early stage in processing.
There are several effective means of removing moisture from natural gas. One of the most common methods is glycol dehydration. Another method is cryogenic drying, and a third possibility is to remove water from the gas by molecular sieve. But regardless of the dehydration method, it is essential to make control measurements of the dried gas to ensure that the drying equipment is functioning properly and the remaining water vapour is only present at or below acceptable levels. Exactly what constitutes this acceptable level is defined by regulations, guidelines, and contracts.
A device for measuring water content is known as a hygrometer. Over the years, a number of methods and technologies have been developed to make this measurement, but the first successful method to achieve the necessary level of accuracy, reliability and repeatability was the ‘Bureau of Mines Tester’, a chilled-mirror device that measures the WDP (Figure 1). At any given pressure, the WDP temperature corresponds to the amount of water vapour present. Using a chilled-mirror instrument, the dew point can be measured directly. This earliest chilled-mirror device, developed in the late 1930s, was operated manually and required the use of a refrigerant gas to chill the mirror while the operator observed the surface of a stainless-steel mirror that was exposed to a sample of natural gas flowing across it. A thermometer was embedded in the mirror in such a way as to give accurate information about the mirror temperature. The operator used this to identify the temperature when water began to condense as the mirror was cooled. It was also possible to observe when the water began to evaporate as the mirror slowly warmed to the ambient temperature after the refrigerant gas was removed.
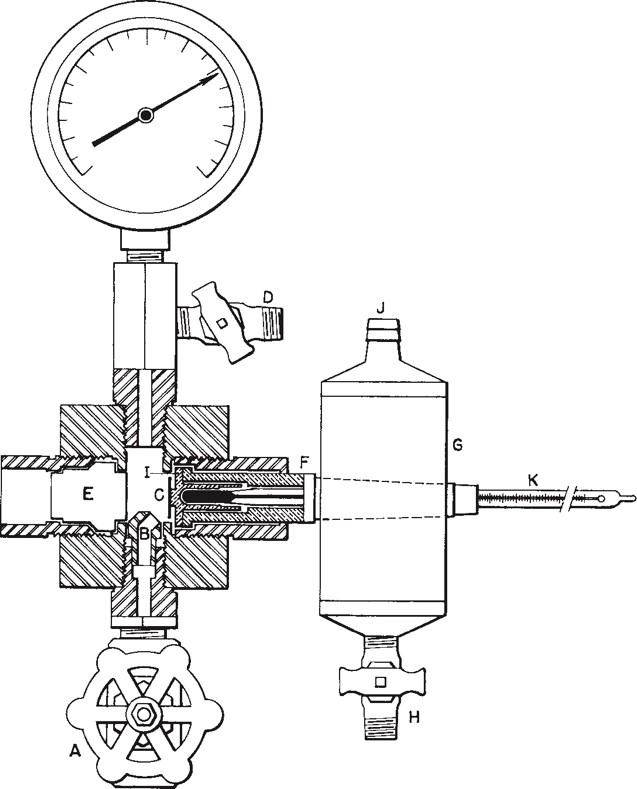
Figure 1. The Bureau of Mines Dew Point Tester (1938) was the first device that made possible accurate repeatable measurement of the water dew point in hydrocarbon gases. It was subsequently used to monitor other condensates as well.
This device required skill and practice to use well, but it did provide good results that were acceptable at the time.
A dew point is a physical property of the gas, so measuring it directly with a chilled mirror is a first-principle measurement, which is considered to be of the highest metrological value. As a result, a chilled mirror analyser still provides the best dew point measurement.
Knowing the dew point makes it possible to determine the humidity in terms of parts per million (ppm), milligrams per cubic meter (mg/m3), pounds per million standard cubic feet (lbs/million ft3) or any other units desired or deemed appropriate. Moreover, once the dew point is known for one pressure it is possible to find the dew point at a different pressure, as this is a simple calculation.
Since those first manual devices were developed, there have been significant advances in both automatic and manual incarnations of chilled-mirror technology (Figure 2). While the underlying principles of operation have never changed, specifically a temperature-controllable mirror, a light source to reveal condensation, and a means of observing this condensation – either visually or with some type of photo sensor – one major improvement was introduced with the development of automated devices: controlled heating of the mirror (Figure 3). In modern chilled-mirror devices, both manual and portable, the mirror is heated as well as cooled by means of a thermoelectric battery, a so-called ‘Peltier’ element. During heating and cooling, the temperature is monitored electrically and temperature changes are controlled by a microprocessor. The ability to heat the mirror as well as cool it made two important advances in functionality possible.

Figure 2. The Hygrovision-mini is a modern manual hygrometer that operates on the same principles as the Bureau of Mines Tester, but incorporates modern electronics and optics to enhance performance.
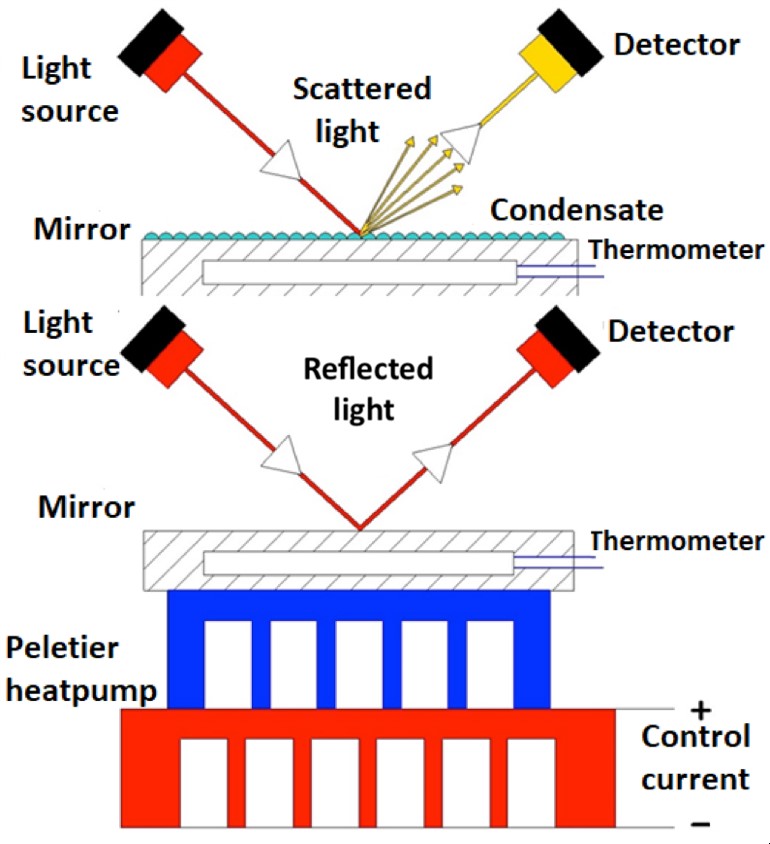
Figure 3. A schematic view of the essential components of an automatic chilled mirror hygrometer.
In the first place, the ability to heat the mirror resulted in the complete elimination of all condensates between each measurement cycle. This is achieved by heating the mirror to over 40 ̊C and maintaining that temperature long enough to ensure that all condensates have evaporated. In this way, each measurement starts with a clean mirror.
And secondly, by heating the mirror in the same controlled manner as it is cooled, accurate measurement of the evaporation temperature can be directly compared to measurement of the condensation temperature. In other words, the rate of mirror temperature change is the same going up and going down. The reason that this is a relevant improvement becomes clear when it is understood that the dew point is not merely the water condensation temperature, but is in fact the equilibrium temperature at which water is both condensing and evaporating at equal rates. In a chilled-mirror device, the condensation temperature observed will be slightly below the dew point as the mirror must be cooler than the dew point for observable condensation to form (Figure 4). To compensate for this, the evaporation temperature is also measured. In this case, the temperature must be slightly above the dew point for evaporation to be observable. By finding the mean temperature between the condensation and evaporation temperatures, an accurate dew point can be established based on direct measurements.

Figure 4. Water condensation on the surface of the chilled mirror of a manual hygrometer. (Photographed through the microscope of a Hygrovision dew point analyser).
This is the procedure described in both the ASTM D1142-95 ‘Standard Test Method for Water Vapor Content of Gaseous Fuels by Measurement of Dew-Point Temperature’ and the ISO 6327 ‘Gas analysis – Determination of the water dew point of natural gas – Cooled surface condensation hygrometers’ 1,2. There is even reference to “observation of the temperature at which the [water condensation] deposit appears and disappears, as indicated by a thermometer” in the original paper describing the Bureau of Mines apparatus3.
In the absence of a measured evaporation temperature, it is not possible to establish by direct measurement how near the observed condensation temperature is relative to the actual dew point. Devices that do not measure the evaporation temperature are required to introduce an assumed correction value to compensate for the difference between the actual dew point and the measured condensation temperature. But this value is not based on any continuous direct measurement information. These devices are not in conformance with the requirements of the relevant standards.
Although other technologies for measuring the moisture content in natural gas have been developed, they do have drawbacks, and actual chilled-mirror instruments are still considered to be the most accurate devices, providing reliable and repeatable measurements.
Liquid-state hydrocarbons
Like water, hydrocarbons can also condense into a liquid state under the right temperature and pressure conditions. As is the case with water, liquid state hydrocarbons in the pipeline can cause a number of problems. In particular, when these liquids are carried along in the gas flow, they form bullet-like slugs that are extremely damaging to equipment such as gas-fired turbines and compressors.
Unprocessed natural gas often has a number of heavier hydrocarbons that condense at relatively high temperatures and require removal before the gas can be fed into the distribution grid. Fortunately, once removed, these components can be sold for their own value. As is the case with moisture removal, it is necessary to ensure that the processing equipment is working efficiently and effectively removing all of the components necessary to ensure that the gas quality reaches the appropriate standard. The HCT is a reliable indicator of how well the heavier hydrocarbons have been removed and chilled-mirror devices are also used for this measurement. In fact, chilled-mirror is the only available technology that can measure this temperature directly.
Similar to the WDP measurement, the surface of a mirror is cooled until hydrocarbon condensates form. Unlike water, however, hydrocarbons do not form little droplets when they condense. Instead, they form a flat reflective film when they condense onto a chilled surface (Figure 5). Additionally, hydrocarbons are not a single component but many different components. Though in many respects similar, in terms of condensation temperature, each hydrocarbon behaves and interacts differently. As a result, the way that the hydrocarbon matrix in a given natural gas behaves cannot be predicted. This adds to the accuracy issue when calculating this value based on a chromatographic analysis, and it is simply not possible to calculate the HCT from one pressure to another, unlike the WDP.

Figure 5. Hydrocarbon condensation on the surface of the chilled mirror of a manual hygrometer. (Photographed through the microscope of a Hygrovision dew point analyser).
Because of the negative impacts associated with liquid hydrocarbons in the pipeline, it is advisable to know the temperature that the gas must remain above in order to prevent potential hydrocarbon liquids (PHCLs) from precipitating out of the gaseous phase. But this presents another problem. Whereas the WDP simply increases as the pressure rises, the HCT only increases to a certain pressure. Above this pressure the HCT actually goes down again.
Since it is not possible to recalculate the HCT from one pressure to another, it is important to consider measuring this temperature at the pressure that corresponds to the highest HCT. This pressure is known as the cricondentherm. For example, if the operating pressure is 70 bar and the HCT is measured at this pressure, it would not be possible to anticipate what the temperature would be downstream of a pressure reduction facility, where the gas pressure is reduced at say 30 bar. The HCT will almost certainly be higher at 30 bar than at 70 bar, but by how much is not knowable without directly measuring it. Each hydrocarbon gas or gas mixture has a unique cricondentherm.
The CONG Prima online and Hygrovision portable series of instruments manufactured by the Vympel company enable both the WDP and HCT to be measured using one measurement cell in a single instrument
Conclusion
Quality control is a crucial aspect of maintaining the modern natural gas infrastructure and supply. A major requirement of that quality control is the installation of instrumentation to monitor specific characteristics and components of the gas. Two of the most crucial continuous measurements are for the WDP and the HCT.
Measurement of the WDP and HCT, despite some similarities, are fundamentally different. Both measurements are critical to maintaining a healthy natural gas infrastructure and delivering high quality natural gas. But, whereas finding the WDP gives clear information about one specific component of the gas, measuring the HCT provides information about the gas matrix as a whole.
Finding the WDP provides information about how much water vapour is present in the gas, and at what temperature that water vapour will begin to condense into liquid.
Finding the HCT provides information about the temperature, at the measured pressure, at which hydrocarbon liquids will begin to form and about the presence, or not, of heavier hydrocarbons unaccounted for in the most common type of gas chromatograph analysis.
Each of these measurements requires technology fine-tuned to the requirements of the component(s) being monitored. It is an interesting coincidence that chilled-mirror technology is appropriate for both measurements, and it is noteworthy that there are devices that can accurately and reliably measure both values with one instrument.
References:
1. ASTM International, Standard D 1142-95, ‘Standard Test Method for Water Vapor Content of Gaseous Fuels by Measurement of Dew-Point Temperature’, (2000).
2. International Organization for Standardization, ISO 6327, ‘Gas analysis – Determination of the water dew point of natural gas – Cooled surface condensation hygrometers’, (1981).
3. United States Department of the Interior R.I. 3399, (May 1938).
